Molecular Immunology:大豆过敏原表位研究
发布日期:2020-04-30
原标题:用过敏患者的血清来确定大豆球蛋白表位,可以发现许多豆类和坚果常见的共享表位
•对位于大豆球蛋白六聚体暴露表面的大多数表位进行三维建模
•与其它植物11S贮藏蛋白序列比较,发现一个显性表位和多个共享表位
•不同植物家族的贮藏蛋白之间通常会共享常见的“热点”表位
越来越多的西方饮食中增加了大豆的食用量,但最近有研究表明,普通人群中大豆过敏的患病率约为0.5%,在儿童中这一比例高达13%。
本研究采用2D凝胶电泳技术,研究6例大豆过敏成人血清与大豆蛋白免疫球蛋白E(IgE)结合的情况。使用包含大豆球蛋白A2和A3亚基主序列的合成肽来绘制IgE结合区域,同时利用生物信息学软件来定位在大豆球蛋白六聚体模型上的假定表位。
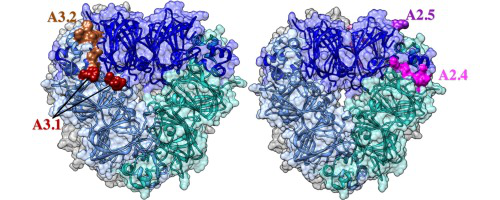
我们通过筛选大豆过敏患者的血清,确定了主要贮藏蛋白Gly m 6的线性IgE结合表位。然后进一步在三维硅胶模型中进行定位分析,并与其他植物贮藏蛋白表位进行比较。我们也利用网络软件来探究准确预测表位的能力,但结果参差不齐。最终我们在大豆球蛋白A3(A3.1–A3.3)和A2(A2.1–A2.6)亚基中总共鉴定出9个IgE结合表位。
除一名患者的血清与四种不同的A2表位结合外,大部分患者的血清IgE仅与一或两种表位结合。9个结合表位中的两个表位(A3.2和A2.4)与先前确定的来自其他植物的11S球蛋白“热点”表位区重叠。大多数表位被预测可暴露在大豆球蛋白六聚体的三维模型表面。对大豆球蛋白和其他植物球蛋白进行氨基酸序列比对揭示了一个显性“热点”表位区,且该“热点”区属于已报道的四个“热点”区。这项研究可能有助于对大豆过敏免疫治疗和诊断的未来发展研究。
延伸阅读
Molecular Immunology Volume 70, February 2016,Pages 125-133
https://doi.org/10.1016/j.molimm.2015.12.008
Abstract:
Highlights •
The IgE-binding B-cell epitopes of two similar soybean glycinin subunits (Gly m 6) are described.
• Three-dimensional modeling of the glycinin hexamers located most epitopes on the exposed surfaces.
• Comparison with other plant 11S storage protein sequences revealed one dominant and several shared epitopes.
• Common epitope hot spots are often shared between plant storage proteins from diverse families.
Abstract
Soybean consumption is increasing in many Western diets; however, recent reviews suggest that the prevalence of soy allergy can be as high as 0.5% for the general population and up to 13% for children. The immunoglobulin-E (IgE) binding of sera from six soy-sensitive adult human subjects to soybean proteins separated by 2D gel electrophoresis was studied.
Synthetic peptide sets spanning the mature glycinin subunit A2 and A3 primary sequences were used to map the IgE-binding regions. Putative epitopes identified in this study were also localized on glycinin hexamer models using bioinformatics software.
We identified linear IgE-binding epitopes of the major storage protein Gly m 6 by screening individual soy-sensitive patient sera. These epitopes were then further analysed by 3D in silico model localization and compared to other plant storage protein epitopes. Web-based software applications were also used to study the ability to accurately predict epitopes with mixed results. A total of nine putative IgE-binding epitopes were identified in the glycinin A3 (A3.1–A3.3) and A2 (A2.1–A2.6) subunits.
Most patients' sera IgE bound to only one or two epitopes, except for one patient's serum which bound to four different A2 epitopes. Two epitopes (A3.2 and A2.4) overlapped with a previously identified epitope hot spot of 11S globulins from other plant species. Most epitopes were predicted to be exposed on the surface of the 3D model of the glycinin hexamer. Amino acid sequence alignments of soybean acidic glycinins and other plant globulins revealed one dominant epitope hot spot among the four reported hot spots. This study may be helpful for future development of soy allergy immunotherapy and diagnosis.
All Authors:
Hanaa Saeed Christine Gagnon Elroy Cober Steve Gleddie


——浙大迪迅 译
本研究主要描述了两种相似大豆球蛋白亚基(Gly m 6)结合IgE的B细胞表位•对位于大豆球蛋白六聚体暴露表面的大多数表位进行三维建模
•与其它植物11S贮藏蛋白序列比较,发现一个显性表位和多个共享表位
•不同植物家族的贮藏蛋白之间通常会共享常见的“热点”表位
越来越多的西方饮食中增加了大豆的食用量,但最近有研究表明,普通人群中大豆过敏的患病率约为0.5%,在儿童中这一比例高达13%。
本研究采用2D凝胶电泳技术,研究6例大豆过敏成人血清与大豆蛋白免疫球蛋白E(IgE)结合的情况。使用包含大豆球蛋白A2和A3亚基主序列的合成肽来绘制IgE结合区域,同时利用生物信息学软件来定位在大豆球蛋白六聚体模型上的假定表位。
我们通过筛选大豆过敏患者的血清,确定了主要贮藏蛋白Gly m 6的线性IgE结合表位。然后进一步在三维硅胶模型中进行定位分析,并与其他植物贮藏蛋白表位进行比较。我们也利用网络软件来探究准确预测表位的能力,但结果参差不齐。最终我们在大豆球蛋白A3(A3.1–A3.3)和A2(A2.1–A2.6)亚基中总共鉴定出9个IgE结合表位。
除一名患者的血清与四种不同的A2表位结合外,大部分患者的血清IgE仅与一或两种表位结合。9个结合表位中的两个表位(A3.2和A2.4)与先前确定的来自其他植物的11S球蛋白“热点”表位区重叠。大多数表位被预测可暴露在大豆球蛋白六聚体的三维模型表面。对大豆球蛋白和其他植物球蛋白进行氨基酸序列比对揭示了一个显性“热点”表位区,且该“热点”区属于已报道的四个“热点”区。这项研究可能有助于对大豆过敏免疫治疗和诊断的未来发展研究。

延伸阅读
Molecular Immunology Volume 70, February 2016,Pages 125-133
[IF:3.188]
Using patient serum to epitope map soybean glycinins reveals common epitopes shared with many legumes and tree nutshttps://doi.org/10.1016/j.molimm.2015.12.008
Abstract:
Highlights •
The IgE-binding B-cell epitopes of two similar soybean glycinin subunits (Gly m 6) are described.
• Three-dimensional modeling of the glycinin hexamers located most epitopes on the exposed surfaces.
• Comparison with other plant 11S storage protein sequences revealed one dominant and several shared epitopes.
• Common epitope hot spots are often shared between plant storage proteins from diverse families.
Abstract
Soybean consumption is increasing in many Western diets; however, recent reviews suggest that the prevalence of soy allergy can be as high as 0.5% for the general population and up to 13% for children. The immunoglobulin-E (IgE) binding of sera from six soy-sensitive adult human subjects to soybean proteins separated by 2D gel electrophoresis was studied.
Synthetic peptide sets spanning the mature glycinin subunit A2 and A3 primary sequences were used to map the IgE-binding regions. Putative epitopes identified in this study were also localized on glycinin hexamer models using bioinformatics software.
We identified linear IgE-binding epitopes of the major storage protein Gly m 6 by screening individual soy-sensitive patient sera. These epitopes were then further analysed by 3D in silico model localization and compared to other plant storage protein epitopes. Web-based software applications were also used to study the ability to accurately predict epitopes with mixed results. A total of nine putative IgE-binding epitopes were identified in the glycinin A3 (A3.1–A3.3) and A2 (A2.1–A2.6) subunits.
Most patients' sera IgE bound to only one or two epitopes, except for one patient's serum which bound to four different A2 epitopes. Two epitopes (A3.2 and A2.4) overlapped with a previously identified epitope hot spot of 11S globulins from other plant species. Most epitopes were predicted to be exposed on the surface of the 3D model of the glycinin hexamer. Amino acid sequence alignments of soybean acidic glycinins and other plant globulins revealed one dominant epitope hot spot among the four reported hot spots. This study may be helpful for future development of soy allergy immunotherapy and diagnosis.
All Authors:
Hanaa Saeed Christine Gagnon Elroy Cober Steve Gleddie
2020-1-17 Article
创建过敏性疾病的科研、科普知识交流平台,为过敏患者提供专业诊断、治疗、预防的共享平台。

 杭州浙大迪迅生物基因工程有限公司
杭州浙大迪迅生物基因工程有限公司

